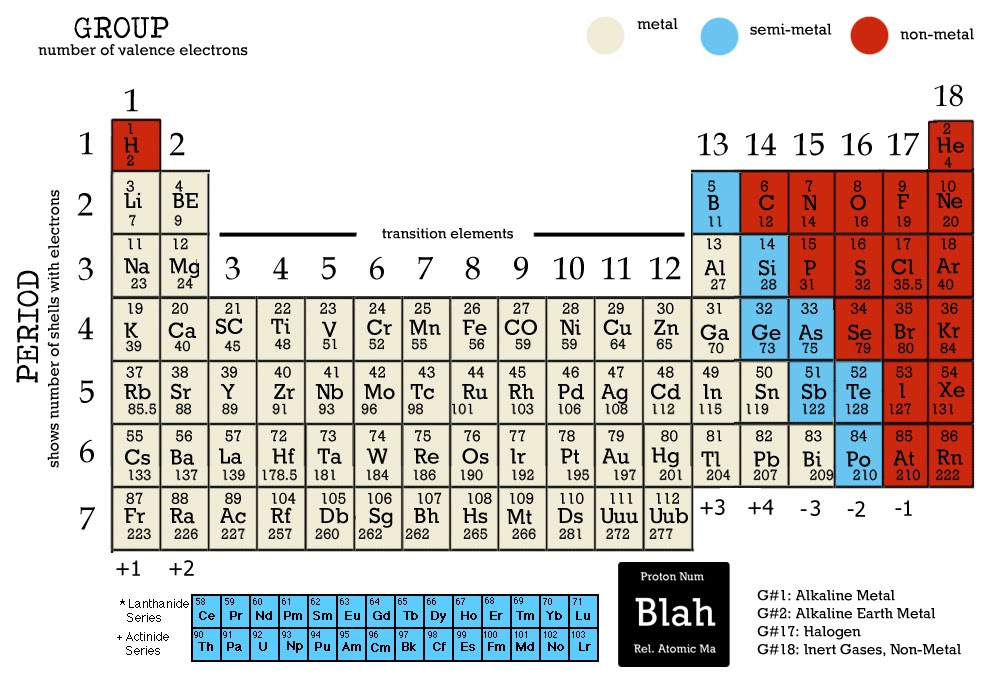
In Chapter 1, we described Dalton’s theory that each chemical compound has a particular combination of atoms and that the ratios of the numbers of atoms of the elements present are usually small whole numbers. (8 atoms)(15.9994 amu/atom) = 127.9952 amuįormula mass of Ca 3(PO 4) = 310.177 2amu Match the descriptions (a)(e) with the following terms: atomic weight, atomic mass, mass number, atomic number, molar mass. If you know the molecular weight, you know the molar mass. The units for molecular mass are given in atomic mass units while the units for molar mass are give in grams per mol. (2 atoms)(30.973761 amu/atom) = 6.0474 amu Each atom, therefore, can be assigned both an atomic number (the number of protons equals the number of electrons) and an atomic weight (approximately equaling. Periodic table with charges Periodic table with electron configuration Periodic table with density Metals, nonmetals & metalloids Elements’ data. Molecular weight is the mass of one molecule of a substance while the molar mass is the mass of one mole of a substance. The Maxwell-Boltzmann speed distribution curve for N 2 at 25✬ is shown below. Graphing this equation gives us the Maxwell-Boltzmann distribution of speeds. When you use the molar mass for a calculation, you round the final. 
In real cases, it is usually fine to calculate molar mass by using the atomic masses shown on your periodic table. The reason for calculating a molar mass is to use it in a real calculation. From their positions on the periodic table, will N and S form a molecular. However, that may be unnecessary - and even undesirable. In the mid-19 th century, James Maxwell and Ludwig Boltzmann derived an equation for the distribution of molecular speeds in a gas. The atomic masses in Table 3.5 Selected Atomic Masses of Some Elements are. Use the atomic mass of sulfur from the periodic table and the following atomic masses to determine whether these data are accurate, assuming that these are the only isotopes of sulfur: 31. Distributions of the Value of Molecular Speed. \right ) \right ]=310.177\ amu \notag \) Atoms Using a mass spectrometer, a scientist determined the percent abundances of the isotopes of sulfur to be 95.27 for 32 S, 0.51 for 33 S, and 4.22 for 34 S.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
